New Cosentyx® treatment can clear skin of patients with psoriasis
While there is no cure for psoriasis, an autoimmune skin disease, the science of psoriasis therapies is evolving at a very rapid pace. The recent Cosentyx® approval heralds a new era for psoriasis treatment at SkinCare Physicians’ Psoriasis Treatment Center near Boston.
Cosentyx® (secukinumab), a biologic therapy developed specifically for psoriasis, is now available for adults suffering from moderate-to-severe psoriasis. This is the first of several highly anticipated “second generation” biologics to complete rigorous testing and become available to treat this significant life-impacting disease.
Cosentyx® targets interleukin-17 (IL-17), one of the key chemical hormones that are elevated in people who have psoriasis. We now know that psoriasis is driven by underlying and excessive inflammation, and IL-17 is one of the critical entities involved in this process.
With this novel treatment, clear skin may now be a reality for patients suffering from psoriasis. In clinical trials, the success rate was astounding with over 70% of subjects achieving clearance or almost clearance of their psoriasis within 16 weeks. Common side effects included uncomplicated cold symptoms, headache and mild diarrhea.
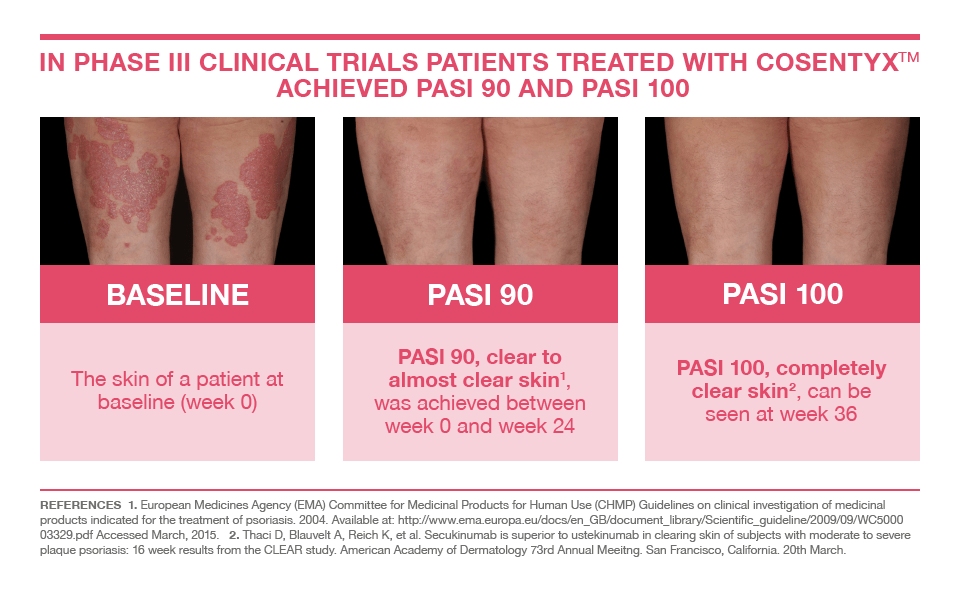
PASI 90 means that patients experienced a 90% or more reduction in their Psoriasis Area and Severity Index (PASI)
Picture courtesy of Novartis Pharmaceuticals Corporation
Cosentyx® joins Enbrel®, Humira®, Stelara® and Remicade ® as biologic options for treating moderate-to-severe psoriasis. The addition of Cosentyx® to our psoriasis treatment armamentarium is a big win for patients still suffering from this disease. The odds are now exceedingly high that any patient who medically is a candidate for these therapies will find a treatment that works for them.
And the good news is that this is just the beginning. There are at least 4 other promising second generation biologic agents for psoriasis currently in their final stages of testing that may see approval in the next 1-3 years. It is an evolving and exciting time, and our Psoriasis Treatment Center in Boston has never been more equipped to help patients manage this disease.
Learn more about psoriasis:
- The heartbreak of psoriasis: Not just a cosmetic nuisance
- Advancements in psoriasis treatment hitting an all-time high



Can I have a tattoo removed using a q-switched ND-YAG Laser while on this medication?
Yes, undergoing laser treatment for tattoo removal is acceptable while receiving Cosentyx.
Can you freeze your eggs while on cosentyx?
There is unfortunately no data for this. I suggest consulting with both your prescribing provider and your OB/GYN
Hello, please, I was just prescribed this medicine for scalp psoriasis and arthritis .
I already have acne, and monthly cystic acne during my Menst. periods which still left scars. I have already tried everything for years for acne.
So my question is: Is there a higher risk due to my previous medical condition that this new medicine will worse the existing condition of the acne?
thanks.
It is unlikely that Cosentyx will have any negative effect on your acne. Acne was not a common side effect seen in the clinical trials for Cosentyx.
I have used Cosentyx for 5 months now. I need to have tooth extraction and implants placed. How should I time the surgery and maybe delay the next cosentyx injection for how long? What is the half life?
Hello, we recommend that you contact your physician directly to get an answer to your question. Regards.
Can a 16 years old receive this treatment, he weights 189lbs
Cosentyx is approved for adults age 18 or older. There are, however, effective psoriasis therapies that are available for those less than 18. Enbrel is approved for psoriasis ages 4 and higher and Stelara for ages 12 and higher. We suggest a consultation with a dermatologist if interested in finding out more about these options.
Is it ok to have tattoos done on my skin?? – I have been on Corsentyx for 10 months. Prior to corsentyx I had severe Psoriasis all over body, face and scalp and 6 weeks after starting Corsentyx (Apr 2018) I became completely clear and still am upto writing this comment.
Hi,
There are 2 potential concerns: first, injury to the skin can sometimes induce a new plaque of psoriasis. Second, as cosentyx can increase your risk of infection, you may run a higher risk of skin infection with the tattoo procedure. So these are relative contraindications, they are not absolute.
When using Cosentyx – which I have been on now for 6 weeks – can I have cosmetic fillers applied to my face?
i.e. Collagen [Cosmoderm & Cosmoplast], Restylane and Juvederm [hydolonic acid based fillers]
and Botox?
I am having trouble finding an answer to these questions.
Thank you.
Yes, receiving fillers is acceptable when on biologic therapies, including Cosentyx. Be certain that your skin has been cleaned prior to the injection to reduce any risk of skin infection.
Could this medication heal acne?
Hello, Cosentyx is approved solely for psoriasis and psoriatic arthritis. It has not been studied for acne.